Clinical evidence & safety
Proliv™Rx was studied in a highly challenging patient population – individuals who had previously failed to achieve adequate response to antidepressant therapy.
It is the first and only at-home neuromodulation therapy to demonstrate significant efficacy in a prospective, randomized controlled clinical trial with patients that have historically been difficult to treat and underserved.
Indication for Use
The Proliv™Rx System provides focal external Combined Occipital and Trigeminal Afferent Stimulation (eCOT-AS) treatment. It is intended as an adjunctive treatment for Major Depressive Disorder (MDD) in adults who failed to achieve satisfactory improvement from at least one previous antidepressant medication, for patients use at home or in clinic. The device is a prescription only device.

The MOOD Study: landmark clinical evidence in major depressive disorder
The MOOD study, led by leading psychiatrists in the United States with Dr. Linda Carpenter of Brown University as the lead principal investigator, evaluated the safety and efficacy of Proliv™Rx in adults with Major Depressive Disorder (MDD) who had an inadequate response to antidepressant medication.
Key study characteristics
- Adults with MDD and inadequate response to 1–4 antidepressants
- Randomized, double-blind, sham-controlled design
- Conducted at 13 clinical sites
- 8-week double-blind treatment period (active vs. placebo), followed by an additional 8-week open-label phase with active treatment.
- Participants self-administered the Proliv™Rx therapy at home, completing two 40-minute sessions per day, 5–7 days per week.
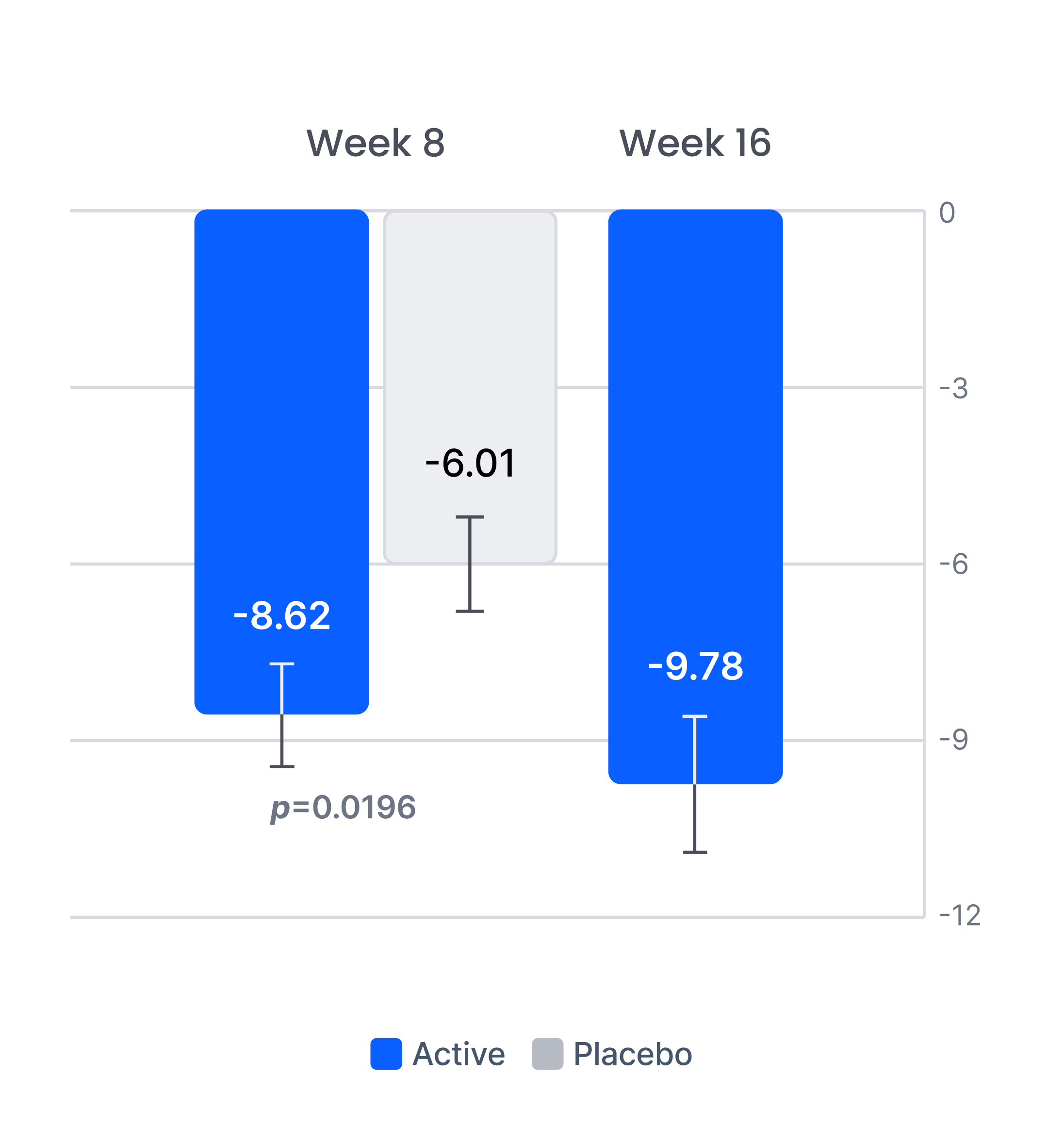
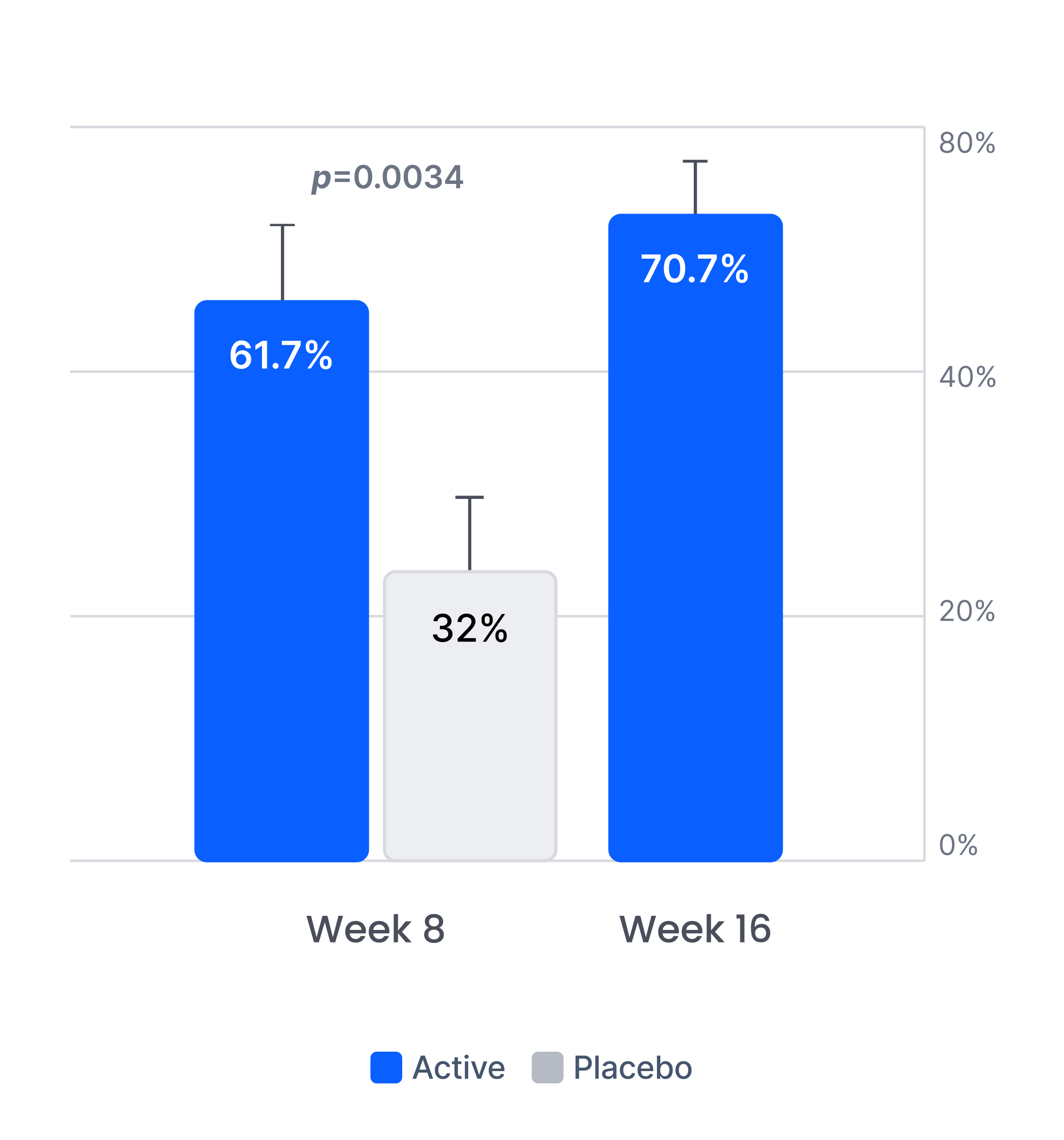
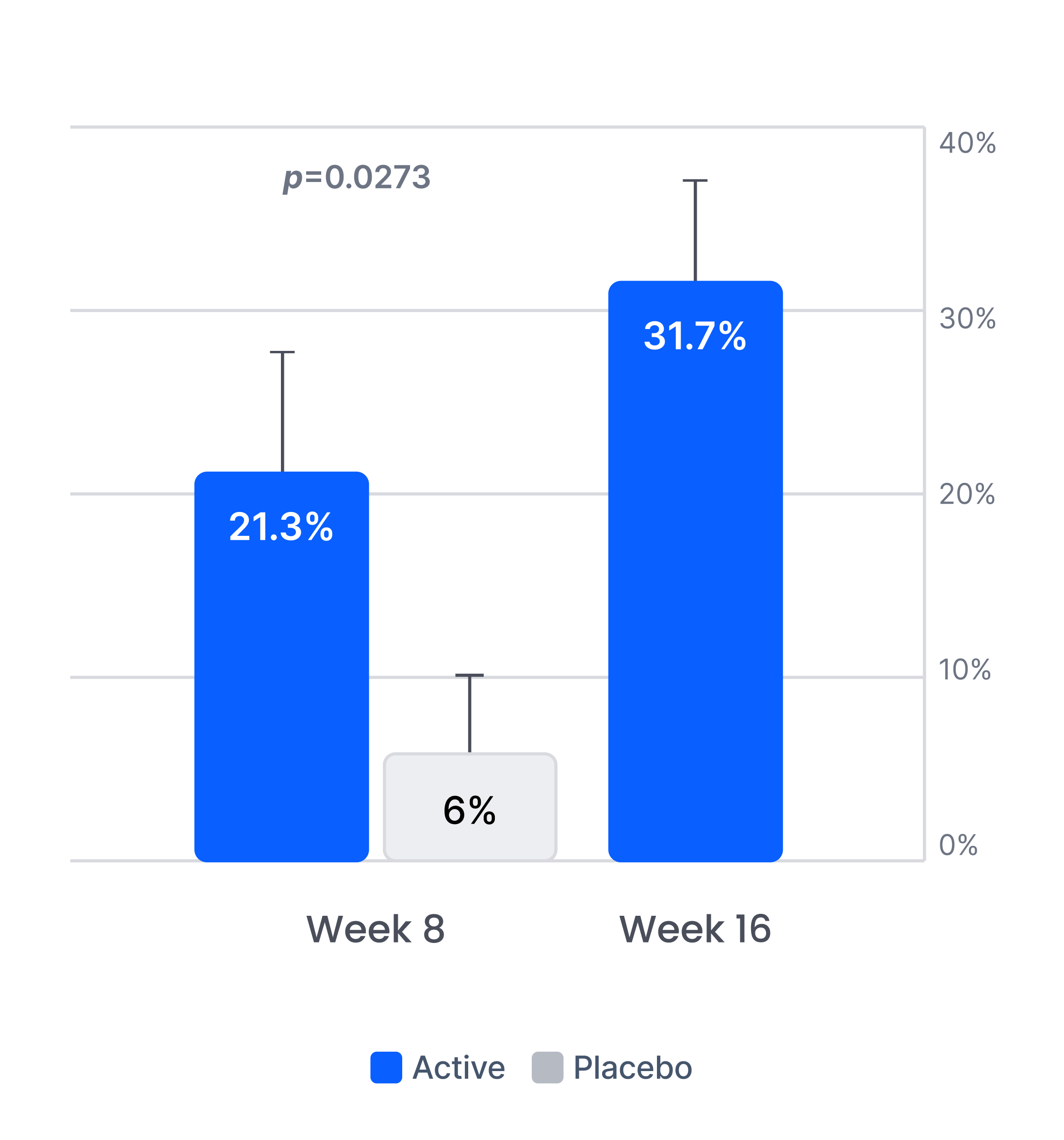
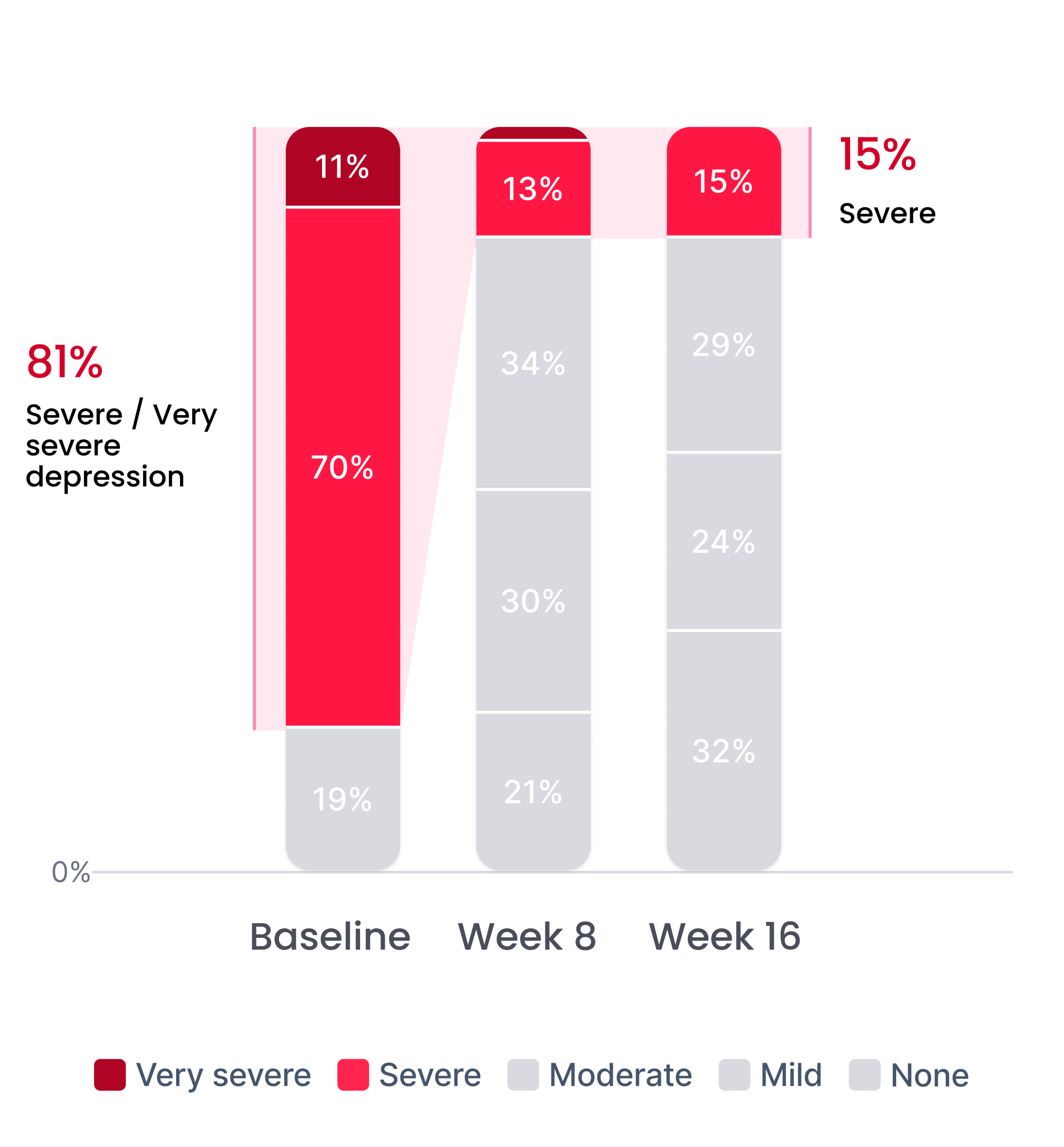
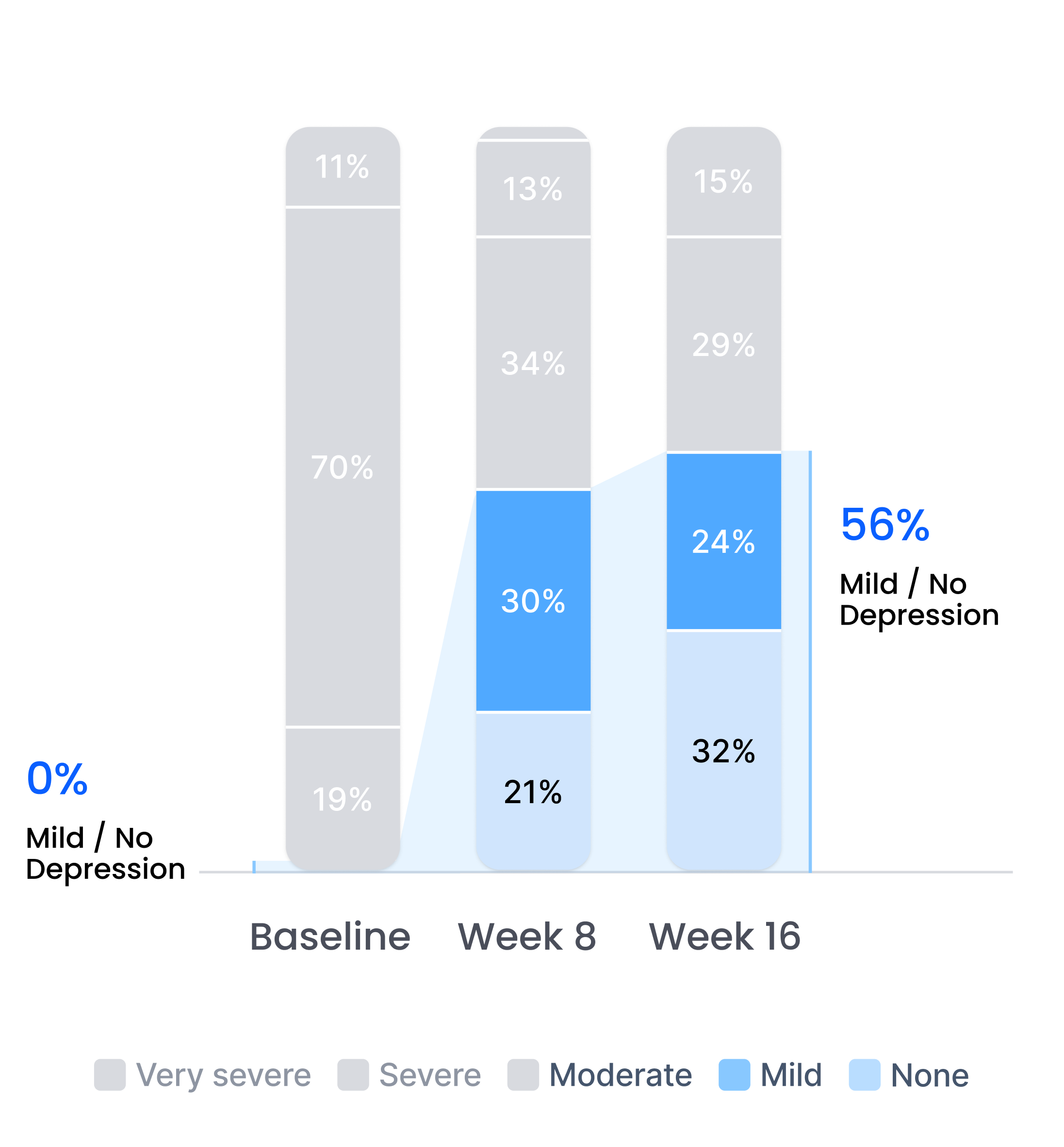
Efficacy in patients with prior inadequate response to antidepressants
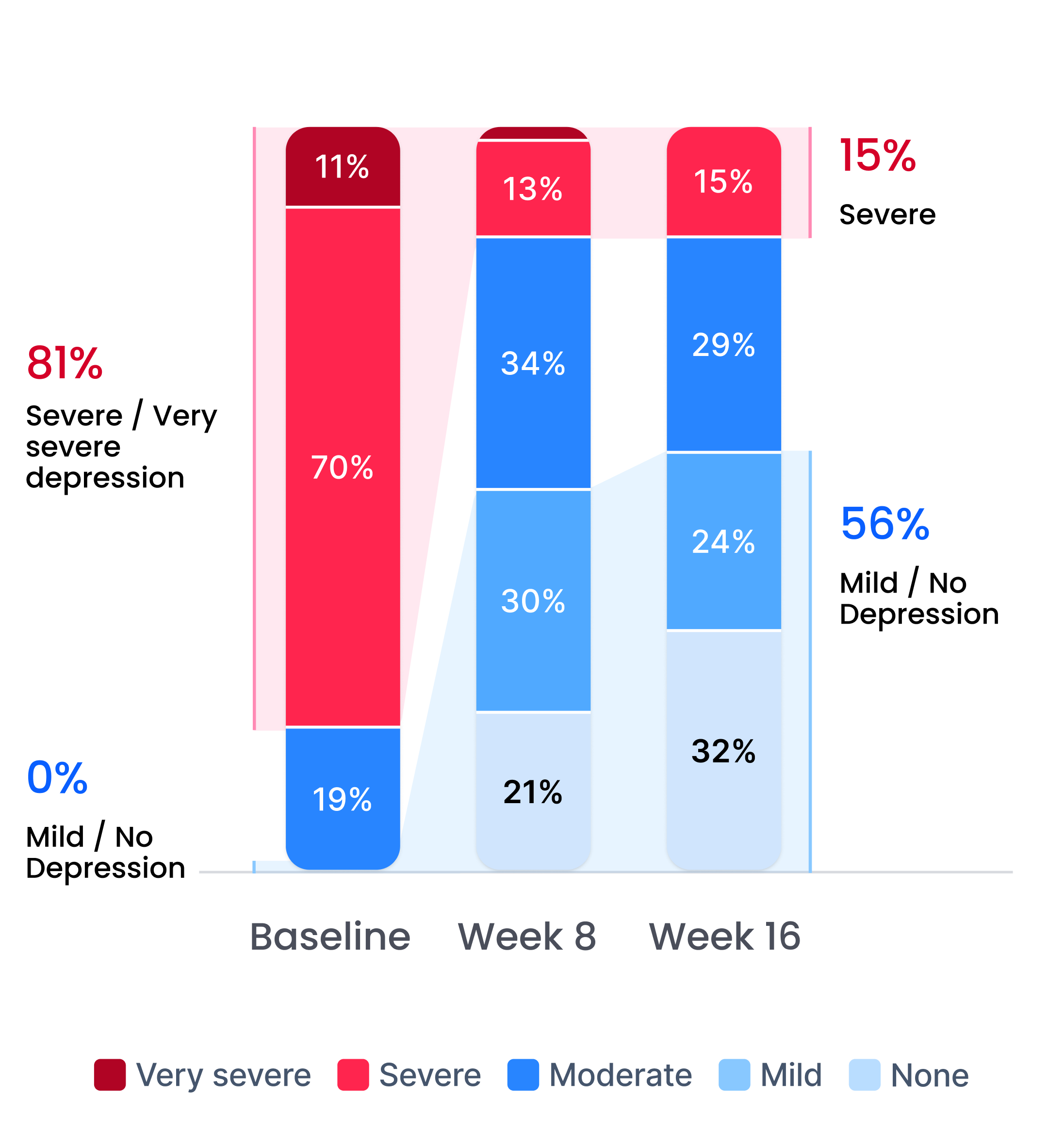
Improvement in depression severity level
Depression severity levels: Very Severe, Severe, Moderate, Mild, No Depression. After completion of a 16-week therapy program.
Proliv™Rx: key clinical outcomes after antidepressant failure
Favorable safety profile
Proliv™Rx was well tolerated in clinical studies, with no device-related unanticipated serious adverse events reported. Observed adverse events were generally mild to moderate in severity and transient in nature. The most commonly reported adverse events included scalp numbness, localized skin reactions at the electrode sites, and headache.
Clinical Resources & Publications